
What you eat can eliminate you: bloodmeal sources and mosquito fitness
Multiple hosts, multiple impacts: the role of vertebrate host diversity in shaping mosquito life history and pathogen transmission
Abstract
Recommendation: posted 01 June 2023, validated 02 June 2023
Santiago-Alarcon, D. (2023) What you eat can eliminate you: bloodmeal sources and mosquito fitness . Peer Community in Infections, 100079. https://doi.org/10.24072/pci.infections.100079
Recommendation
Diptera-borne pathogens rank among the most serious health threats to vertebrate organisms around the world, particularly in tropical areas undergoing strong human impacts – e.g., urbanization and farming –, where social unrest and poor economies exacerbate the risk (Allen et al. 2017; Robles-Fernández et al. 2022). Although scientists have acquired a detailed knowledge on the life-history of malaria parasites (Pacheco and Escalante 2023), they still do not have enough information about their insect vectors to make informed management and preventive decisions (Santiago-Alarcon 2022).
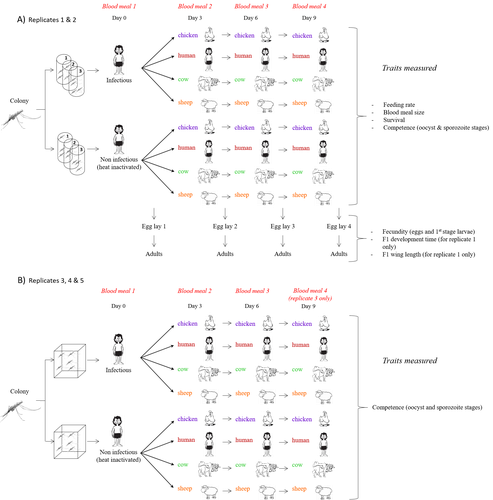
In this sense, I am pleased to recommend the study of Vantaux et al. (2023), where authors conducted an experimental and theoretical study to analyzed how the diversity of blood sources (i.e., human, cattle, sheep, and chicken) affected the fitness of the human malaria parasite – Plasmodium falciparum – and its mosquito vector – Anopheles gambiae s.l.
The study was conducted in Burkina Faso, West Africa. Interestingly, authors did not find a significant effect of blood meal source on parasite development, and a seemingly low impact on the fitness of mosquitoes that were exposed to parasites. However, mosquitoes’ feeding rate, survival, fecundity, and offspring size were negatively affected by the type of blood meal ingested. In general, chicken blood represented the worst meal source for the different measures of mosquito fitness, and sheep blood seems to be the least harmful. This result was supported by the theoretical model, where vectorial capacity was always better when mosquitoes fed on sheep blood compared to cow and chicken blood. Thus, the knowledge generated by this study provides a pathway to reduce human infection risk by managing the diversity of farm animals. For instance, transmission to humans can decrease when chickens and cows represent most of the available blood sources in a village.
These results along with other interesting details of this study, represent a clear example of the knowledge and understanding of insect vectors that we need to produce in the future, particularly to manage and prevent hazards and risks (sensu Hoseini et al. 2017).
REFERENCES
Allen T., et al., Global hotspots and correlates of emerging zoonotic diseases. Nat. Commun. 8, 1124. (2017). https://doi.org/10.1038/s41467-017-00923-8
Hosseini P.R., et al., Does the impact of biodiversity differ between emerging and endemic pathogens? The need to separate the concepts of hazard and risk. Philos. Trans. R. Soc. Lond. B Biol. Sci. 372, 20160129 (2017). https://doi.org/10.1098/rstb.2016.0129
Pacheco M.A., and Escalante, A.A., Origin and diversity of malaria parasites and other Haemosporida. Trend. Parasitol. (2023) https://doi.org/10.1016/j.pt.2023.04.004
Robles-Fernández A., et al., Wildlife susceptibility to infectious diseases at global scales. PNAS 119: e2122851119. (2022). https://doi.org/10.1073/pnas.2122851119
Santiago-Alarcon D. A meta-analytic approach to investigate mosquitoes’ (Diptera: Culicidae) blood feeding preferences from non-urban to urban environments. In: Ecology and Control of Vector-borne Diseases, vol. 7 (R.G. Gutiérrez-López, J.G. Logan, Martínez-de la Puente J., Eds). Pp. 161-177. Wageningen Academic Publishers. eISBN: 978-90-8686-931-2 | ISBN: 978-90-8686-379-2 (2022).
Vantaux A. et al. Multiple hosts, multiple impacts: the role of vertebrate host diversity in shaping mosquito life history and pathogen transmission. bioRxiv, ver. 3 peer-reviewed and recommended by Peer Community in Infections (2023). https://doi.org/10.1101/2023.02.10.527988
The recommender in charge of the evaluation of the article and the reviewers declared that they have no conflict of interest (as defined in the code of conduct of PCI) with the authors or with the content of the article. The authors declared that they comply with the PCI rule of having no financial conflicts of interest in relation to the content of the article.
ANR grant no. 11-PDOC-006-01
Evaluation round #1
DOI or URL of the preprint: https://doi.org/10.1101/2023.02.10.527988
Version of the preprint: 2
Author's Reply, 17 May 2023
Decision by Diego Santiago-Alarcon, posted 08 Apr 2023, validated 08 Apr 2023
I agree with both reviewers that this is a very interesting study that provides novel and highly valuable information. However, along with both reviewers I have comments that need to be considered before the article is recommended.
Please address all the comments raised by the reviewers and myself, and whenever you disagree provide a rational behind your decision. My comments are attached in a Word document.
Download recommender's annotationsReviewed by Francisco C. Ferreira, 01 Mar 2023
The study by Vantaux and colleagues evaluated a broad spectrum of mosquito fitness traits that may be impacted by human infections with Plasmodium falciparum and host choice in subsequent blood meals. Through extensive experimental challenges using four different blood sources in sequential feedings, the authors found that Plasmodium-infected mosquitoes had lower feeding rates compared to uninfected mosquitoes, an effect observed for all four blood types in the first feeding after Plasmodium exposure. Infection status did not affect any other trait, which were largely influenced by the host species of the blood source. Additionally, the blood source did not affect Plasmodium development within the mosquito. These results provided essential parameters to model vectorial capacity in multi-host environments.
The introduction is comprehensive and provides a clear rational behind the study. The manuscript is fairly well-written. However, bellow I have addressed some parts that could be further clarified. The laboratory methods used to evaluate the fitness traits are reliable, and the statistical analyses performed are robust .
The authors used field isolates of P. falciparum and lab populations of An. coluzzii that were frequently replenished with wild-caught F1 mosquitoes, making the experimental set-up similar, but not identical to "natural" conditions. This is a major contribution of this study, but it would be important that the authors provide evidence that the replenishment of lab colonies with wild mosquitoes if sufficient to remove inbreeding effects that may affect mosquito-Plasmodium interactions. I am saying this because a substantial portion of the first paragraph of the 'Discussion' is dedicated to comparisons between natural and non-natural experiments. Although this was addressed in the "study limitations" section, the authors could tone down the assumption that they used a natural system.
It would be important to include the effect of blood meal size on the models evaluating mosquito survival, fecundity, development time and F1 wing length since the amount of blood ingested may play a role in these traits.
It is unclear why the authors kept mosquitoes in cages for trials 3-5. Was it for convenience since the traits measured in these trials did not require close observation of individual mosquitoes? In any case, this could be clarified early in the 'Methods' section.
It's very interesting that feeding rate was the only trait affected by Plasmodium infection. However, does the experimental set-up (artificial feeding) allow authors to extrapolate this effect to real-world scenarios? I wonder if it would be better to use feeding-rate data from studies using whole-body to address feeding success. Even studies using immobilized hosts may provide more realistic information compared to the present study that used parafilm membrane for feeding. Using these parameters for the modeling is probably fine, though, since the cross-comparisons among blood types provide the relative impact of each host species.
The authors explain only in the 'Discussion' why they did not sugar-fed mosquitoes in their experiments. It may be worth explaining this in the 'Methods' as well. In the 'Discussion', it would be interesting to see some explanation about how sugar-feeding may alleviate potential fitness costs in infected mosquitoes. Has this been demonstrated before? Ferguson et al 2003 (https://doi.org/10.1111/j.0014-3820.2003.tb01521.x) provide evidence that sugar restriction reduces survival in infected mosquitoes.
L314 - Maybe the authors could clarify which prevalence is represented in the pie charts in Fig. 1, whether it’s the oocyst or sporozoite rates.
L503-504 - What did authors mean by "that by not separating exposed-infected and exposed-uninfected females we were not able to measure them". Mosquito groups exposed to either infected or uninfected blood meals were mixed in the same cup/cage? Please clarify this in the 'Methods' and/or in the 'Discussion'.
https://doi.org/10.24072/pci.infections.100079.rev11Reviewed by anonymous reviewer 1, 30 Mar 2023
The article entitled "Multiple hosts, multiple impacts: the role of vertebrate host diversity in shaping mosquito life history and pathogen transmission" provides valuable information on the effect of blood-meals on the physiology, reproduction, survival and epidemiology of Anopheles coluzzi, particularly in the gonotrophic cycle. The authors acknowledge the limitations of a laboratory study, but provide bases for studies in natural environments. Therefore, I recommend being accepted for publication in this journal.
It is a complete and interesting article, I only have one suggestion:
Figure 3 and Figure 6.- The colors cause me confusion, I recommend that in graphs B and D use gray scale or include the four solid or degraded colors in exposed and unexposed.
Figure 2.- It provides relevant information, but it is small. I recommend separating or enlarging, to facilitate reading.
https://doi.org/10.24072/pci.infections.100079.rev12









